Veeva SiteVault Login is a user-friendly and secure cloud-based platform designed to help life sciences organizations manage clinical trials and regulatory documents across multiple locations. It allows authorized users to access and manage clinical information, trial data, documents, and reports easily and safely in real-time from any device with internet access. Veeva SiteVault Login enhances collaboration and communication within research sites and stakeholders, allowing them to work together in a transparent and streamlined manner. With its advanced features and intuitive interface, Veeva SiteVault Login has become one of the most trusted cloud platforms for managing clinical trials and regulatory compliance in the life sciences industry.
How to Veeva Sitevault Login:
1. Open your preferred web browser and go to the Veeva Sitevault login page.
2. Enter your Username and Password in the appropriate fields.
3. Click on the “Login” button.
4. Upon successful verification of your credentials, you will gain access to your Veeva Sitevault account.
What to do if you forgot your Password or Username:
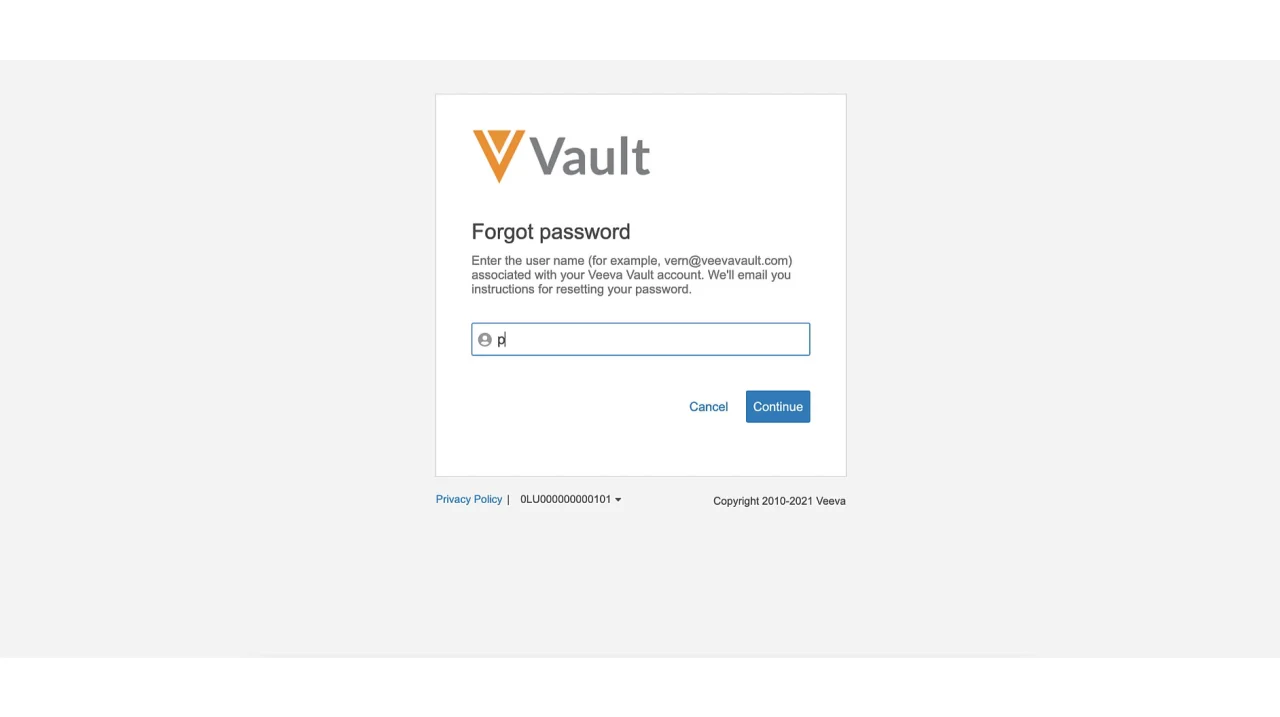
1. Click on the “Forgot Password?” link on the Veeva Sitevault login page.
2. Enter your email address or username associated with your Veeva Sitevault account.
3. Follow the instructions sent to your registered email address to reset your password.
4. If you forgot your username, you can contact Veeva customer support to retrieve it.
5. Once you have reset your password or retrieved your username, you can log in to your Veeva Sitevault account.
Keyword 1: Veeva Vault Login
Veeva Vault Login refers to the process of accessing the Veeva Vault platform, which is designed to help life sciences companies manage their content, data, and workflow. With Veeva Vault Login, authorized users can access the platform securely from any location with an internet connection. To use Veeva Vault Login, users must have a valid username and password, which are provided by their organization’s Veeva Vault administrator. Once logged in, users can perform a variety of tasks, including uploading, viewing, and sharing documents and data, setting access controls, and tracking workflows.
Keyword 2: Veeva Vault Site Login
Veeva Vault Site Login is a related concept to Veeva Vault Login. It refers specifically to the process of logging in to the Veeva Vault Site platform, which is designed for clinical trial sponsors and CROs to manage study site information and documentation. With Veeva Vault Site Login, authorized users can access the platform securely and perform a variety of tasks related to site management, such as adding and editing site information, collecting and reviewing regulatory documents, and monitoring site activity.
Keyword 3: Veeva Vault eTMF Login
Veeva Vault eTMF Login is another related concept, referring to the process of accessing the electronic trial master file (eTMF) module within the Veeva Vault platform. The eTMF is designed to help organizations manage documentation related to clinical trials, with features such as document indexing, version control, and audit trails. With Veeva Vault eTMF Login, authorized users can access the eTMF securely and perform a variety of tasks, such as uploading and organizing documents, reviewing and approving documents, and generating reports.
Keyword 4: Veeva Vault CRM Login
Veeva Vault CRM Login is a related concept, referring to the process of accessing the customer relationship management (CRM) module within the Veeva Vault platform. This module is designed to help life sciences companies manage their customer interactions, with features such as contact management, account management, and activity tracking. With Veeva Vault CRM Login, authorized users can access the CRM module securely and perform a variety of tasks, such as updating customer information, scheduling appointments, and logging sales calls.
Keyword 5: Veeva Vault Training Login
Veeva Vault Training Login is the process of accessing the training module within the Veeva Vault platform. This module is designed to help life sciences companies train their employees and partners on various aspects of the Veeva Vault platform and related software. With Veeva Vault Training Login, authorized users can access the training module securely and participate in a variety of training activities, such as watching videos, taking quizzes, and interacting with trainers and peers. The training module is an essential component of ensuring that users are able to use Veeva Vault effectively and efficiently.
FAQs related to Veeva Sitevault Login:
1. What is Veeva Sitevault Login?
Veeva Sitevault Login is a secure platform for clinical research professionals to access and manage clinical trial data. It helps researchers to collaborate and share information securely across multiple organizations.
2. How do I access Veeva Sitevault?
To access Veeva Sitevault, you need to have valid login credentials provided by your organization. Once you have the login details, you can open the Veeva Sitevault login page and enter your username and password to access the platform.
3. What if I forget my Veeva Sitevault password?
If you forget your Veeva Sitevault password, you can reset it by clicking on the ‘Forgot Password’ link on the login page. You will receive an email with a link to reset your password. Follow the instructions in the email to reset your password.
4. What if I forget my Veeva Sitevault username?
If you forget your Veeva Sitevault username, you can contact your organization’s administrator or Veeva support to retrieve your username.
5. Can I access Veeva Sitevault from anywhere?
Yes, you can access Veeva Sitevault from anywhere as long as you have an internet connection and a compatible web browser.
6. Is Veeva Sitevault secure?
Yes, Veeva Sitevault is a highly secure platform that is compliant with industry-leading security standards. It uses advanced encryption and multi-factor authentication to protect your data from unauthorized access, hacking, and cyber threats.
7. What types of data can I access on Veeva Sitevault?
Veeva Sitevault allows you to access a wide range of clinical trial data, including study protocols, patient records, lab results, adverse events, and other relevant information.
8. Can I share data with external partners through Veeva Sitevault?
Yes, Veeva Sitevault allows you to share data securely with external partners, including contract research organizations, regulatory agencies, and other stakeholders. You can control the access and permissions for each user or group of users to ensure that they only see the data they are authorized to access.
9. How do I report a technical issue or problem with Veeva Sitevault?
If you encounter any technical issues or problems with Veeva Sitevault, you can contact your organization’s administrator or Veeva support for assistance. They will help you diagnose and resolve the issue as quickly as possible.
Getting Started with Veeva SiteVault
Conclusion
In conclusion, the Veeva SiteVault login portal is a powerful tool for clinical trial management. With its user-friendly interface, secure login credentials, and robust features, it enables clinical trial stakeholders to easily access study data and track progress.
However, as our reliance on digital technology continues to increase, it is becoming more and more important for us to be vigilant about privacy and security concerns. We must ensure that our login credentials are kept safe and confidential, and that we are taking every necessary precaution to protect sensitive data from unauthorized access.
Ultimately, the Veeva SiteVault login portal represents just one piece of a much larger puzzle when it comes to clinical trial management. As we continue to work towards greater transparency, efficiency, and patient-centricity in the world of clinical research, we must remember that technology is merely a tool – it is up to us to use that tool wisely and ethically. With that in mind, let us approach our work with intention, purpose, and a deep commitment to the health and wellbeing of patients everywhere.